 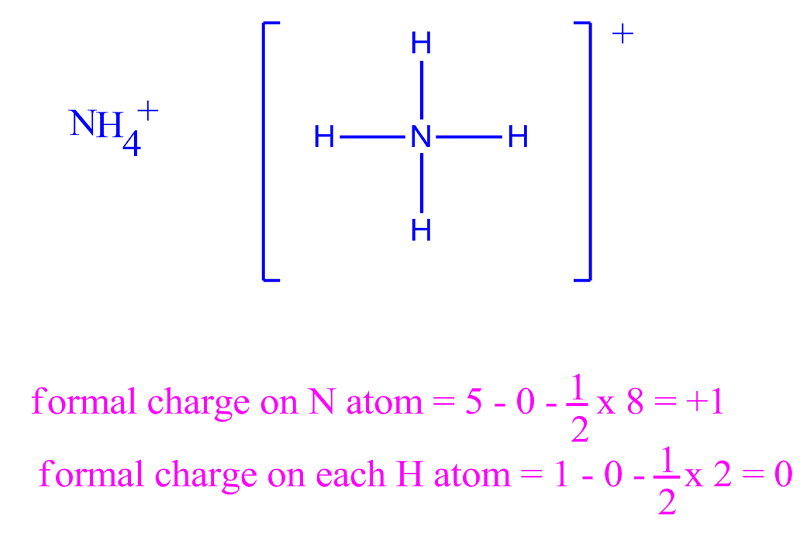
Each Cl atom now has seven electrons assigned to it, and the I atom has eight. We assign lone pairs of electrons to their atoms. A double bond between two atoms is shorter (and stronger) than a single bond between the same two atoms. We divide the bonding electron pairs equally for all ICl bonds: Step 2. If nitrite ions do indeed contain a single and a double bond, we would expect for the two bond lengths to be different. In order to calculate the formal charges for NO3- well use the equationFormal charge of valence electrons - nonbonding val electrons - bonding ele. Brackets surround this structure, and there is a superscripted negative sign. Calculating Formal Charge from Lewis Structures Assign formal charges to each atom in the interhalogen molecule BrCl 3. The right structure shows an oxygen atom with two lone pairs of electrons double bonded to a nitrogen atom with one lone pair of electrons that is single bonded to an oxygen atom with three lone pairs of electrons. Calculating Formal Charge: Definition & Formula - Quiz & Worksheet Video Quiz Course Try it risk-free for 30 days Instructions: Choose an answer and hit 'next'. To find the formal charge of an atom, subtract the number of non-bonding electrons and half the number of bonded electrons from the number of its valence. Calculate the formal charge for each atom in the carbon monoxide molecule: Answer: C 1, O +1. 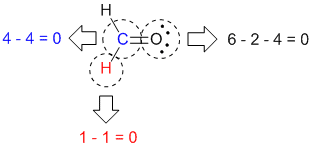
Brackets surround this structure, and there is a superscripted negative sign. To obtain the of an atom, we start by counting the number of valence electrons for the neutral atom, and then subtract from it the number of electrons that it owns ( i.e. One can calculate the formal charges for any given atom with the help of the following formula: F. These charges help in knowing if the given structure of the molecule is stable or not. The left structure shows an oxygen atom with three lone pairs of electrons single bonded to a nitrogen atom with one lone pair of electrons that is double bonded to an oxygen with two lone pairs of electrons. Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. The electrons involved in the N–O double bond, however, are in different positions: The one with the least/lowest formal charges is the ideal structure. 
\) can have two possible structures with the atoms in the same positions. An atom in a molecule should have a formal charge of zero to have the lowest energy and hence the most stable state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
